In previous editions of this newsletter, we’ve discussed some of the research strategies being pursued to enable a severely injured brain to regrow healthy and functional brain tissue. Today, we will explore progress toward replacing lost or damaged brain tissue with new brain matter to support the recovery of lost capabilities.
At first, it seems implausible that new brain tissue can be created and successfully transplanted into someone who has survived a devastating brain injury. While it is now possible to replace a severely damaged liver with a portion of a healthy liver, and the transplanted fragment can grow into a complete, fully functional liver, it is hard to imagine that the same can be done with brain tissue. Several barriers must be overcome if damaged brain tissue is ever to be replaced by new and functional brain tissue.
The first challenge is to identify a suitable source for replacement brain tissue. In recent years, it has become possible to transform cells found in our blood or snippets of skin into pluripotent stem cells. These induced pluripotent stem cells (iPSCs) can be reprogrammed into many different types of cells, including neurons. The reprogrammed iPSCs are called derived neurons. It is now possible, even common, to create vast numbers of these derived neurons, which can be grown in cell culture and attain the electrical properties of the neurons in an intact brain.
If the derived neurons created from an individual are then transplanted back into that individual, these transplanted neurons will not be rejected as foreign since they contain the identity markers of the same individual.
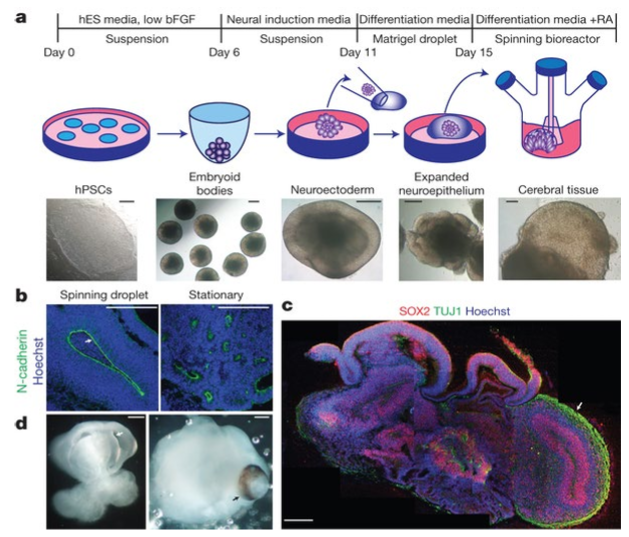
Ten years ago, Lancaster and colleagues¹ demonstrated that it is possible to grow these derived neurons from a 2-dimensional sheet of neurons into an organoid, a small ‘mini-brain,’ a structure that is a few millimeters in diameter. These organoids develop to have many cell types and features found in a living whole brain. Organoids can only live in cell culture for a matter of months, though. They’re not connected to a circulatory system, so their growth is limited.
Nevertheless, the creation of self-organizing organoids demonstrates a crucial principle – derived neurons contain all the information necessary to create a complete brain.
Once organoids were successfully created, teams of researchers began efforts to transplant these organoids into living brains. The typical experimental system used for these experiments is a brain-injured rodent with a depleted immune system. Experiments focused on creating conditions so that a human organoid can be transplanted into such an animal. The goal was to see if organoids can connect to the host animal. Can these transplanted organoids grow new blood vessels? Can the neurons grow functional connections into the brains of the host? Recently, there have been convincing demonstrations that human brain organoids can not only connect to the host animal,² but these transplanted tissues can become functionally active in conjunction with the host’s brain tissue. It has been unequivocally demonstrated that visual stimulation of the mouse causes activation of the transplanted human organoid tissue. Functional synapses (connections) between the human transplanted organoid tissue and the mouse brain are present.³ It appears that the transplanted organoids are ‘seeing’ the mouse’s visual inputs!
Transplanted organoids can even develop new long projections and compensate for infarcted brain tissue and cause functional recovery.⁴ Since it is now possible, in principle, to create new brain cells that can be grown into transplantable tissue that will functionally connect to the host brain, what other challenges will have to be addressed if this approach is to become practical for a brain-injured person? One challenge will be to create a suitable anatomical site for the transplantation of new tissue. Severe brain injuries result in a tangle of scar, debris, and partially active islands of residual brain tissue. The surgical techniques necessary to permit transplantation of brain tissue will have to be developed. But even if it becomes possible to implant new, viable tissue into a person’s brain and support its growth and proliferation, the next challenge will be to optimize the ability of the new tissue to form new connections without disrupting the synaptic activity of the host’s intact brain. There is a growing science and pharmacology of ‘synaptic plasticity.’ What drugs or stimulation patterns can cause neurons to respond to stimulation and create new functional connections?
Optimizing the plasticity of a transplanted organoid in the brain, while ensuring that the existing synaptic connections in the rest of the brain are not disrupted, is a complex challenge. The process will involve careful modulation of various factors to promote the integration of the organoid into the existing neural network without causing adverse effects. A number of strategies are being considered. Some are focused on applying specific growth factors and neurotrophins to the organoid. Others are focused on carefully targeted electrical stimulation, genetic engineering to modify organoids to activate or suppress plasticity genes, or local administration of drugs that modulate synaptic plasticity. This challenge en route to clinical use of organoid tissue is not yet solved. In general terms, we know that newly transplanted tissue must be ‘programmed’ to become functionally useful and integrated into pre-existing brain tissue. One of the fundamental ideas of brain science is that ‘neurons which fire together, wire together…’. New connections between neurons are created and reinforced by experience. If new brain tissue is introduced into an adult brain, the challenge will be to create conditions that optimize the capability of the new tissue to become programmed by external stimulation while not disrupting the stable connections of the pre-existing brain. We’ll likely learn, over the years to come, about techniques to enhance the programmability of the new tissue without causing loss of function in preexisting tissue. One aspect of programming new brain tissue will involve the interfacing of computational devices and algorithms to translate external ‘real-world’ signals into neuronal stimulation patterns and to train new neuronal tissue to control motor behaviors. “Closed loop” devices will, inevitably, accelerate the training of newly implanted brain tissue. The progress in organoid biology allows us to envision a (hopefully) not-too-distant future where new neuronal tissue grown from a person’s own induced stem cells can be transplanted into a damaged brain. This transplantable tissue, grown from iPSC-derived organoids, will contain the information to ‘self-organize’ and will not proliferate uncontrollably. This tissue will be (in this future scenario) introduced into a surgically optimized environment. It will mature, differentiate, grow new blood vessels, and create connections to preexisting functional brain tissue. A pharmacologic formula to optimize new synaptic programming will be developed. At that point, the transplanted DLF organoid tissue will need to be programmed by experience and by biomechanical prostheses to restore lost functions.
An entirely new field of bioengineering is just now emerging. It is called “organoid intelligence“5 we’ll devote a future column to this topic, but, in essence, this is the study of techniques used to train and program organoids to analyze signals and direct outputs. Biomechanical methods to program organoids will inevitably be applied to help newly engrafted tissue become functionally useful. To recap… It is now possible to envision a realistic path to a future in which severe brain injuries will be treated by the transplantation of healthy neurons derived from a person’s own tissues and stimulated to grow, differentiate, connect, and learn. The learning will be achieved by putting the new tissue into a maximally adaptive (‘plastic’) state and stimulating it with biomechanical systems. There is, of course, a long way to go until these concepts are perfected and clinical trials are possible. Very substantial financial, technical, and intellectual resources will be required. It is encouraging that the quest to heal a damaged brain is becoming a bioengineering challenge, not a basic science challenge, and engineering challenges can be addressed by focus and resources.
It is the goal of the DLF to raise awareness of the paths toward healing the severely damaged brain, identify strategies to advance the required technologies and focus the resources required to do the work necessary to bring life-transforming therapies to patients.
References
- Lancaster, M. A. et al. Cerebral organoids model human brain development and microcephaly. Nature 501, 373–379 (2013).
- Mansour, A. A. et al. An in vivo model of functional and vascularized human brain organoids. Nat. Biotechnol. 36, 432–441 (2018).
- Wilson, M. N. et al. Multimodal monitoring of human cortical organoids implanted in mice reveal functional connection with visual cortex. Nat. Commun. 13, 7945 (2022).
- Cao, S.-Y. et al. Cerebral organoids transplantation repairs infarcted cortex and restores impaired function after stroke. NPJ Regen Med 8, 27 (2023).
- Smirnova, L. et al. Organoid intelligence (OI): the new frontier in biocomputing and intelligence-in-a-dish. Front. Sci. Ser.1, (2023).